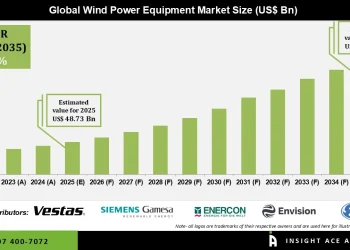
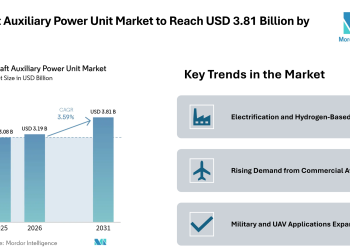
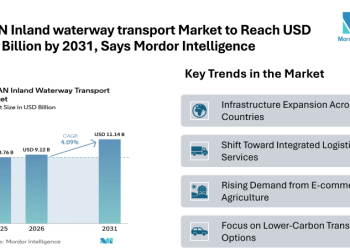
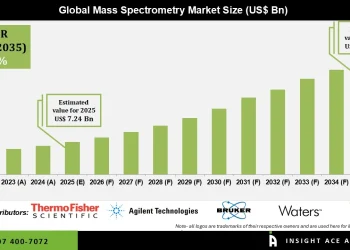
Browse in-depth TOC on “Clinical Trial Supplies Market
237 Tables
47 Figures
242 Pages
Overview of This Study:
This market research study involves the extensive use of secondary sources, directories, and databases to identify and collect information useful for this technical, market-oriented, and financial study of the global clinical trial supplies market. In-depth interviews were conducted with various primary respondents, including key industry participants, subject-matter experts (SMEs), C-level executives of key market players, and industry consultants, to obtain and verify critical qualitative and quantitative information and assess market prospects. The market size was estimated through various secondary research approaches and triangulated with inputs from primary research.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=172240059
Research Methodologies Followed for This Study:
Primary Research:
After acquiring basic knowledge about the global market scenario through secondary research, extensive primary research was conducted. Several primary interviews were conducted with market experts from both the demand side (such as personnel from pharmaceutical & biotechnology companies) and the supply side (such as clinical trial supply companies and pharmaceutical supply companies) across four major regionsNorth America, Europe, the Asia Pacific, and the Rest of the World. Approximately 80% and 20% of primary interviews were conducted with supply-side and demand-side participants, respectively. This primary data was collected through questionnaires, e-mails, online surveys, personal interviews, and telephonic interviews.
Secondary Research:
The secondary sources referred to for this research study include publications from government sources [such as the World Bank, World Health Organization (WHO), and Centers for Disease Control and Prevention (CDC)]; sites like ClinicalTrials.gov, fiercebiotech, and annual reports; SEC filings; press releases; investor presentations; journals; expert interviews; MarketsandMarkets analysis; business magazines and research journals; press releases; and trade, business, and professional associations. Secondary data was collected and analyzed to arrive at the overall size of the global clinical trial supplies market, which was validated through primary research.
Recent Developments:
# In April 2021, Thermo Fisher announced plans to acquire PPD for USD 17.4 billion to enhance its offerings in clinical research services and to support clinical trials
# In April 2021, Parexel collaborated with Veeva Systems to improve their clinical processes for clinical trial delivery and enhance Veevas cloud technology
Request Sample Pages: https://www.marketsandmarkets.com/requestsampleNew.asp?id=172240059
COVID-19 impact on the Clinical Trial Supplies Market
Coronavirus disease 2019 (COVID‐19) is an acute respiratory infectious disease caused by severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2). Largely unknown before the outbreak began in Wuhan (China) in December 2019, COVID-19 has moved from a regional crisis to a global pandemic in just a few weeks. The World Health Organization (WHO) declared COVID-19 as a pandemic on March 11, 2020. There has been a tremendous amount of effort put into developing new treatments and vaccines to cure the infection. Within a few weeks of research, companies started coming up with drugs and treatments to treat the infection.
Browse Adjacent Market: Pharmaceuticals Market Research Reports & Consulting
Related Reports:
Clinical Trial Management System Market by Product (Software, Services), Delivery (Web-hosted, On-premise, Cloud-based), Deployment (Enterprise, On-site), End User (Pharma, Medical Device Manufacturers, CROs) – Global Forecasts to (2022 – 2025)
https://www.marketsandmarkets.com/Market-Reports/clinical-trial-management-systems-market-470.html